San Diego-based biotech company Avacen Medical and CellMark Medical of Gothenburg, Sweden, have formed a strategic partnership to leverage CellMark’s global network across 120 markets to boost sales and marketing of the Avacen-100 Class-II pain relief medical device.
The Avacen-100 recently received the European Union CE (Conformité Européenne) Mark approving it for the treatment of widespread pain associated with fibromyalgia, arthritis, and other conditions by temporarily increasing microcirculation.
The device also has FDA approval as muscular relaxation equipment and Health Canada Medical Device License approval.
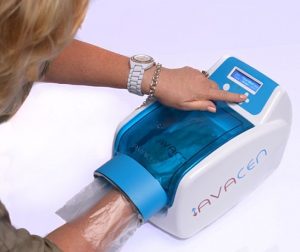
The Avacen-100 uses a non-invasive technique for safely infusing heat into the body’s circulatory system, providing systemic pain relief by using a single point of contact.
The company believes that poor microcirculation is a principal contributing factor in a wide range of health problems including diabetes, hypertension, migraines, vascular disease, atherosclerosis, kidney disease, Alzheimer’s disease, early aging, and others. Avacen estimates that 80% of the population over the age of 40 may have moderately to extremely serious microcirculation problems, and that almost all non-injury related pain can be traced to compromised microcirculation.
Avacen expects four patents issued by the U.S. Patent Office covering innovations incorporated in the Avacen-100 system to allow multiple therapeutic uses for alleviating symptoms associated with various circulatory, neurological, lymphatic, or endocrinal dysfunctions.
Described by its president Niclas Olsson as “a runway for global entrepreneurship,” the employee-owned CellMark Group leverages an expert trading network of 70 offices in 30 countries covering 120 markets working to simplify the export process and minimize associated risks for medical technology manufacturers like Avacen.
Avacen expects its collaboration with CellMark to accelerate global sales and marketing presence by three to five years.
“In addition to CellMark’s existing worldwide distribution channels, they bring vital expertise and relationships which will facilitate rapid regulatory approval of the Avacen-100 in even the most challenging countries throughout the world,” Thomas Muehlbauer, Avacen CEO, said in a press release.
“CellMark Medical was formed to cost effectively bring innovative medical devices to the world that improve quality of life,” Olsson said. “We are pleased to partner with Avacen Medical to bring their unique and effective Avacen-100 to international markets, using our proprietary trade platform, where we have an established network of distributors with broad access to end users.”

